BBC Earth newsletter
BBC Earth delivered direct to your inbox
Sign up to receive news, updates and exclusives from BBC Earth and related content from BBC Studios by email.
Oceans
In the warm seas of the Mediterranean lives a jellyfish with an extraordinarily rare ability – it can rewind its life cycle.
The so-called ‘immortal’ jellyfish, or Turritopsis dohrnii, can somehow reprogramme the identity of its own cells, returning it to an earlier stage of life.
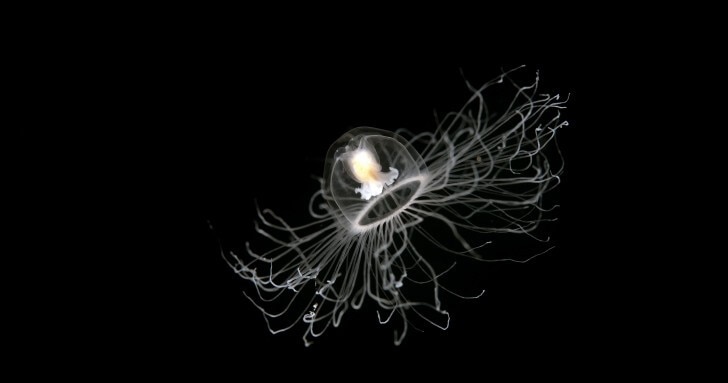
In other words, it can age in reverse and morph from an adult back into a baby.
In the science fiction television series Doctor Who, the programme’s hero periodically transforms into a completely new version of himself. Like Turritopsis dohrnii, the Doctor sometimes does this when he has been badly wounded or would otherwise die.
For the jellyfish, its ability to become a younger version of itself is a spectacular survival mechanism that plays out when it gets old or sick or faces danger. Once the reversal process has been initiated, the jellyfish’s bell and tentacles deteriorate and it turns back into a polyp – a plant-like structure that attaches itself to a surface underwater. It does this partly through a process known as cellular transdifferentiation – when cells change from one type directly into another, producing an entirely new body plan. And it can do it again and again.
A team of researchers recently sequenced a small part of the jellyfish’s DNA. Professor Stefano Piraino at the University of Salento in Italy was involved in the work and is now coordinating a project called PHENIX to better understand the communication between cells in Turritopsis dohrnii. He says the secret of ‘life reversal’ might only be found after the creature’s full genome has been unravelled.
Professor Piraino also notes that the death of the jellyfish has been observed in the lab – perhaps disappointingly, it’s not truly ‘immortal’ after all.
Still, the adaptation remains remarkable and two other jellyfish have recently been discovered to have it, including Aurelia sp.1, native to the East China Sea.
If humans could somehow tap into the transdifferentiation process, could we regenerate? To some extent we already do – scars, bruises and healing after sunburn are all signs of our skin regenerating. We can regrow the tips of our fingers and toes, too.
It was once a popular idea that we become a new person every seven to 10 years as, in that time, all the cells in our body will have died and been replaced. While this myth has been busted, it is true that our cells are continually dying and being replaced.
But as a Time Lord, the Doctor goes through a far more complete transformation process. However, regeneration like his does happen in other animals, though it’s usually limited to just portions of the body. Take salamanders, for instance.
“They are considered the champions of regeneration,” says Dr Maximina Yun at University College London.
They are considered the champions of regeneration”
“Some can regenerate parts of their hearts, their jaws, whole legs and arms – and the tail including the spinal cord.”
The precise mechanism through which salamanders achieve this is not known, but Dr Yun has been experimenting with blastemas – clumps of cells that form when regeneration of a lost body part is triggered at the site of an amputation in salamanders.
She and her colleagues recently found evidence suggesting that salamanders suppress a certain protein, p53, which may contribute to cells taking on a new identity. This would allow cells to develop into the necessary muscle, nerves and bone tissue for a regenerated leg, for example.
It’s hoped that humans may ultimately be able to harness this process for our own benefit (for more on this, see ‘Doctoring ourselves’ on the previous page).
Dr Yun’s team is also investigating the role of the immune system. Once thought to be a potential hindrance to regeneration, she says the presence of immune system cells, macrophages, has now been shown to be ‘essential’ for regeneration. ‘It’s possible that this is the key element,’ she adds.
It’s worth noting that various types of salamander have different means of regenerating. Axolotls, for instance, can trigger the production of stem cells – which can develop into any type of cell – where regeneration is wanted. But newts, when regenerating muscle tissue, use a process called dedifferentiation, in which a specific cell is encouraged to proliferate.
When the Doctor regenerates, he becomes an entirely new person, with a different form and appearance, and perhaps even a different gender. Animals that are able to drastically change their appearance at a moment’s notice are fairly rare, but new examples are being discovered all the time.
Only two years ago, researchers in the Ecuadorian rainforest realised that a certain species of frog, Pristimantis mutabilis, could suddenly change the texture of its skin from rough and spiky to completely smooth in a matter of just a few minutes.
The frog had been known to science for nearly a decade, but this shape-shifting ability – thought to help the frog blend into its environment – had never been documented before.
“It happens so quickly, I think the behaviour just hadn’t been noticed before,” explains Dr Louise Gentle from Nottingham Trent University. “It had been fooling everyone.”
Other animals have long been known to use similar camouflaging tricks, including many species of octopus that can suddenly transform the texture and colour of their bodies to match the surface on which they are resting. It’s not known how this response is triggered. It may be similar to goosebumps on human skin – an involuntary reaction triggered by, for example, low temperatures, explains Dr Gentle.
“But it might be they are recognising their background and consciously making that decision [to transform],” she adds.
Of course, some organisms take on entirely new forms through metamorphosis – the most well-known example being the many caterpillars that form a chrysalis and later emerge as butterflies. But there are some surprising other examples, too. Various species of single-celled amoeba frequently join together as multicellular structures – in other words, they team up to transform. For example, one species, Dictyostelium discoideum, has been found to clump together to form a ‘slug’ when it needs to find food. When the slug has settled on a new feeding ground, it changes form again, into a mature fruiting body that releases spores and begins the life cycle over again.
As more of these examples come to light, Dr Yun observes, “Science is slowly catching up with science fiction.”